Abstract
A multicenter observational study, conducted by the Japanese Society of Hematology (JSH), evaluated the effectiveness and safety of tyrosine kinase inhibitors (TKIs) in newly diagnosed patients with chronic myeloid leukemia -chronic phase (CML-CP). The clinical data of this study were collected using the New Target system, which is an online database that can be easily accessed by physicians.
A total of 506 patients were enrolled from 102 institutions between April 2010 and March 2013; data for 444 patients (median age: 56 [range 18-92] years; 65% male) were analyzed. According to the Sokal score, 201 (45.3%), 178 (40.1%), and 64 patients (14.4%) were classified into the low, intermediate, and high-risk groups. As the first-line therapy, 148 (29.8%) patients received imatinib (IM), 173 (34.8%) received nilotinib (NIL), and 146 (29.4%) received dasatinib (DAS). BCR-ABL1 % was regularly monitored every 3 months in the first year and every 6 months thereafter. TKI plasma trough concentrations were determined with LC-MS/MS at 3, 6, and 12 months.
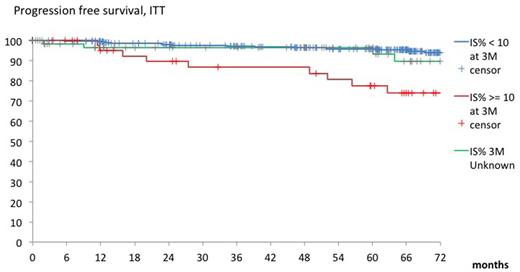
At 60 months of follow up, 5-year progression-free survival (PFS) and overall survival (OS) were 94.5% (95%CI, 91.2-96.5) and 94.8% (95%CI, 91.6-96.9), respectively. The 5-year OS curve was significantly better in patients treated with NIL or DAS than that in patients with IM (IM, 90.9% [95%CI, 83.7-95.0]; NIL, 98.0% [95%CI, 92.2-99.5]; DAS, 96.0% [95%CI, 89.4-98.5]; log-rank test: P =0.0345). An early molecular response (EMR; BCR-ABL1 <10%) was found in 328 patients at 3 months (IM, n=84; NIL, n=127; and DAS, n=117). The 5-year PFS curve was significantly better in patients with EMR than without (EMR, 95.9% [95%CI, 92.6-97.7]; no-EMR, 77.5% [95%CI, 59.8-88.1]; log-rank test: P =0.0002). Additionally, 49 patients (IM, n=9; NIL, n=24; DAS, n=16) achieved deep molecular response (DMR; BCR-ABL1≤0.0032%) by 36 months. The DMR was observed in a higher proportion of patients with EMR (BCR-ABL1<10% at 3 months or BCR-ABL1<1% at 6 months) after TKI treatment (<10%, P =0.0367; <1%, P =0.0068).
Testing for BCR-ABL1mutation was performed in 101 patients. Various BCR-ABL1mutations were identified in 23 patients (IM, n=13; NIL, n=8; and DAS, n=2). T315I mutation was not identified. Exon 8/9 35bp insertion was identified in 14 patients (IM, n=8; NIL, n=5; DAS, n=1). Seven out of these 14 patients achieved DMR; and all patients with exon 8/9 35bp insertion did not progress to accelerated or blast phase .
The achievement of major molecular response (MMR, BCR-ABL1<0.1%) was associated with a higher trough concentration (C0) (DAS mean C02.65 ng/mL in MMR at 6 months vs. 1.98 ng/mL in no-MMR, P=0.0165; NIL mean C0 1,285.3 ng/mL in MMR at 3 months vs. 1,049.8 ng/mL in no-MMR,P=0.0746).
No new safety issues occurred. Among 444 analyzed patients in the safety data set, only 2 patients with NIL had peripheral arterial occlusive disease [grade 1] or acute coronary disease [grade 3]. No patients developed pulmonary arterial hypertension in this study. We measured pleural effusion (PE) in patients with DAS and liver/pancreas dysfunction in patients with NIL, which were the common adverse events. In the DAS arm, 28 patients revealed pleural effusion (grade 1, 3 patients; grade2, 22 patients; grade3, 3 patients). It occurred not only in the first year of treatment but also in the second or third year (median 10.5 [range 1-60] months). However, no statistically significant association was found between pleural effusion and trough concentration of DAS in this study (2.71 ng/mL in patients with PE vs. 2.27 ng/mL in patients without, P =0.1339). In the NIL arm, 17 patients revealed severe liver dysfunction (grade 3-4, n=6), increased bilirubin values (grade 3, n=4), or increased lipase values (grade 3-4, n=7). The adverse events occurred in 3 months in these patients with NIL, except for 4 patients.
In summary, this 5-year Japanese registry using the New Target system demonstrated that TKI treatments in newly diagnosed patients with CML-CP achieved satisfying and trustworthy outcomes.
Takahashi:Bristol-Myers Squibb: Research Funding, Speakers Bureau; Pfizer: Research Funding, Speakers Bureau; Otsuka: Research Funding, Speakers Bureau; Novartis: Research Funding, Speakers Bureau. Kizaki:Nippon Shinyaku,: Research Funding, Speakers Bureau; Novartis: Speakers Bureau; Bristol-Myers Squibb: Research Funding, Speakers Bureau; Celgene: Research Funding, Speakers Bureau. Kawaguchi:Novartis Pharma K.K.: Honoraria, Speakers Bureau; Bristol-Myers Squibb: Honoraria, Speakers Bureau; Pfizer: Honoraria, Speakers Bureau; Alexion: Honoraria, Speakers Bureau. Suzuki:Celgene: Honoraria; Sawai Pharmaceutical: Honoraria; Ohtsuka: Honoraria; MSD: Research Funding; Meiji Seika Pharma: Honoraria; Takeda Pharmaceuticals: Honoraria; Shionogi: Honoraria; Novartis: Honoraria; Mochida Pharmaceutical: Honoraria; Chugai Pharmaceutical: Honoraria; Kyowa-Hakko Kirin: Honoraria; Bristol-Myers Squibb: Honoraria; Sumitomo Dainippon Pharma: Honoraria; Gilead Sciences: Consultancy; MundiPharma: Consultancy; Jazz Pharmaceuticals: Consultancy. Yamamoto:Chugai: Consultancy, Honoraria, Research Funding; Meiji Seika Pharma: Consultancy; Mundipharma: Consultancy, Honoraria; Ono: Consultancy, Honoraria, Research Funding; ARIAD Pharmaceuticals: Research Funding; Bayer: Research Funding; Celgene: Honoraria, Research Funding; Eisai: Honoraria, Research Funding; Gilead Sciences: Research Funding; MSD: Research Funding; Novartis: Honoraria, Research Funding; Solasia Pharma: Research Funding; SymBio: Research Funding; Takeda: Honoraria, Research Funding; AbbVie: Research Funding; Boehringer Ingelheim: Consultancy; Zenyaku: Research Funding; Bristol-Myers Squibb: Honoraria; Kyowa Hakko Kirin: Honoraria; Otsuka: Honoraria; Pfizer: Honoraria; Sumitomo Dainippon: Honoraria; HUYA: Honoraria. Naoe:Nippon Shinyaku Co., Ltd.: Research Funding; Otsuka Pharmaceutical Co., Ltd.: Research Funding; Fujifilm Corporation: Patents & Royalties, Research Funding; Pfizer Japan Inc.: Research Funding; Astellas Pharma Inc.: Research Funding; Toyama Chemical Co., Ltd.: Research Funding. Akashi:MSD: Research Funding; Ono Pharmaceutical: Research Funding; Novartis pharma: Research Funding; Otsuka Pharmaceutical: Research Funding; Chugai Pharma: Research Funding; Astellas Pharma: Research Funding; Eisai: Research Funding; Taiho Pharmaceutical: Research Funding; sanofi: Research Funding; Asahi-kasei: Research Funding; Pfizer: Research Funding; Kyowa Hakko Kirin: Research Funding, Speakers Bureau; Eli Lilly Japan: Research Funding; Celgene: Research Funding, Speakers Bureau; Bristol-Myers Squibb: Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal